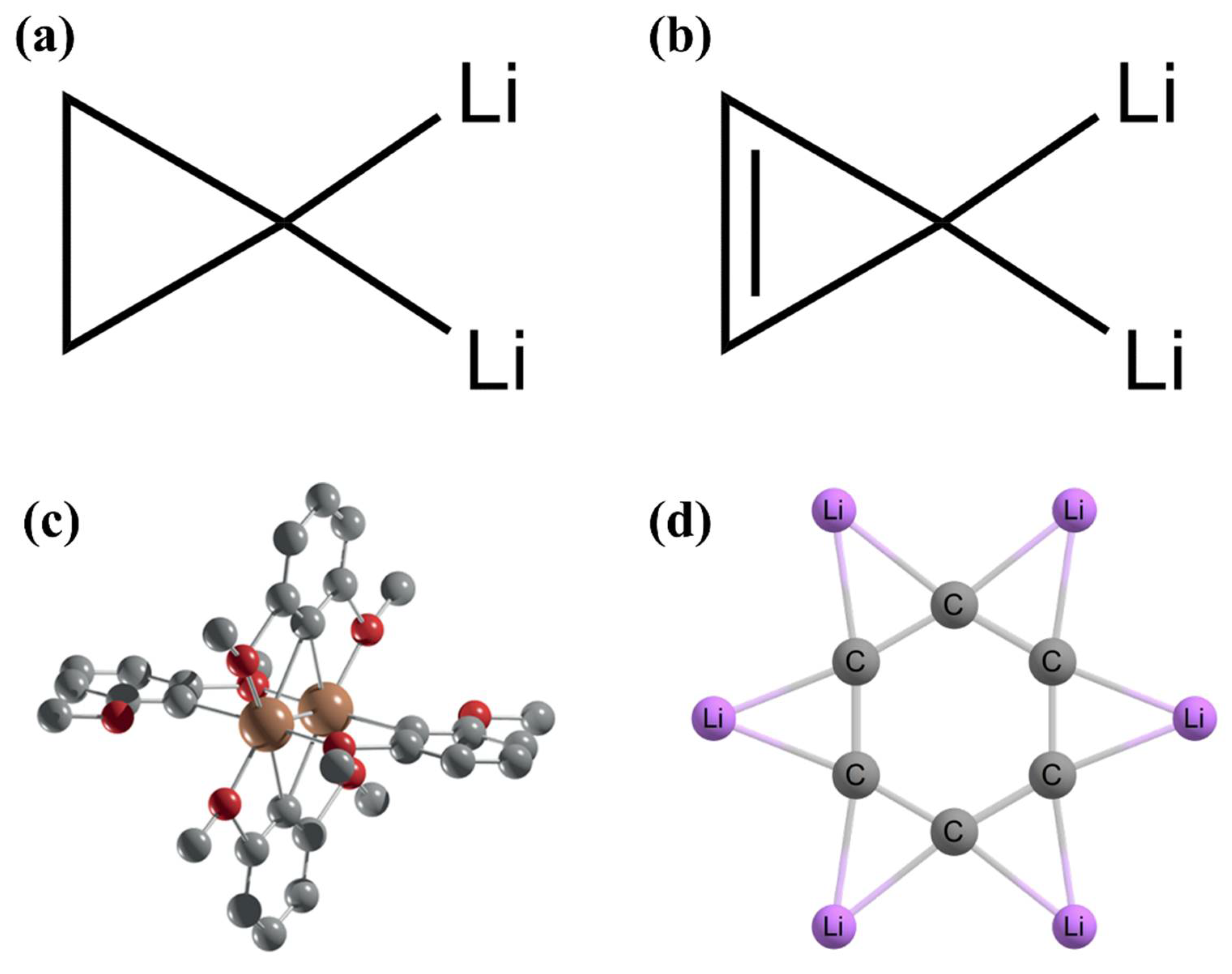
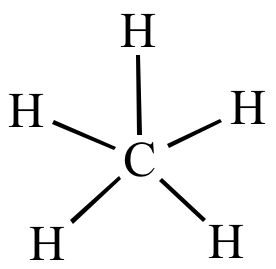
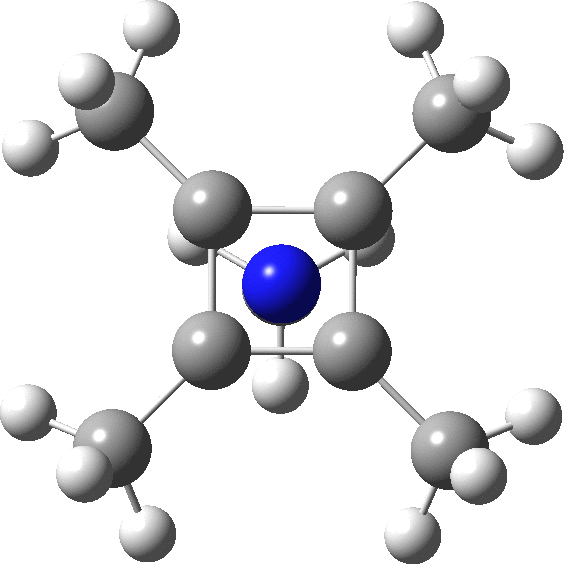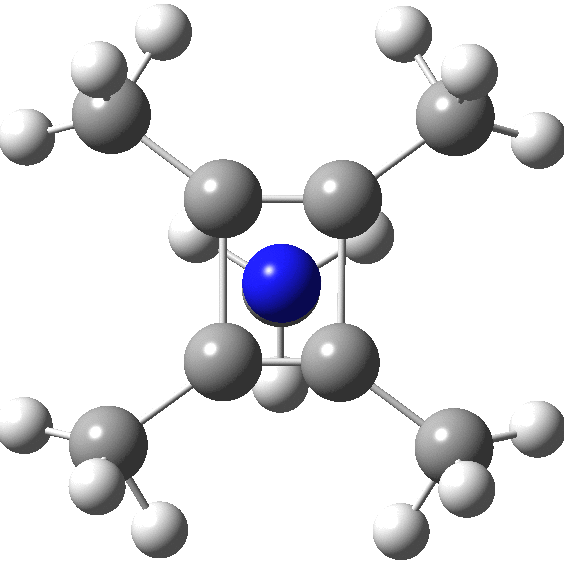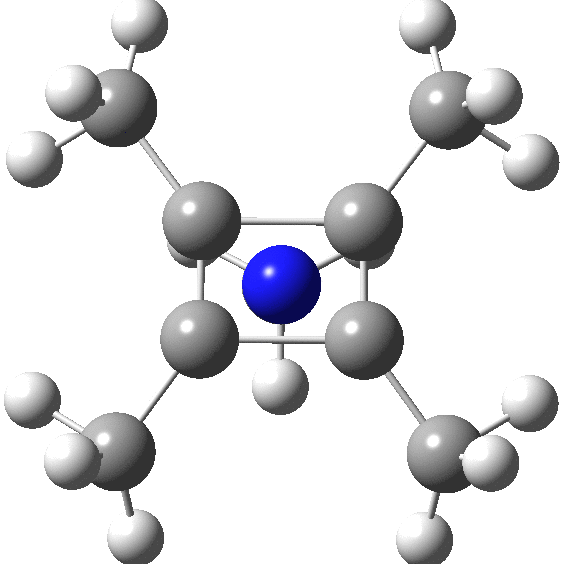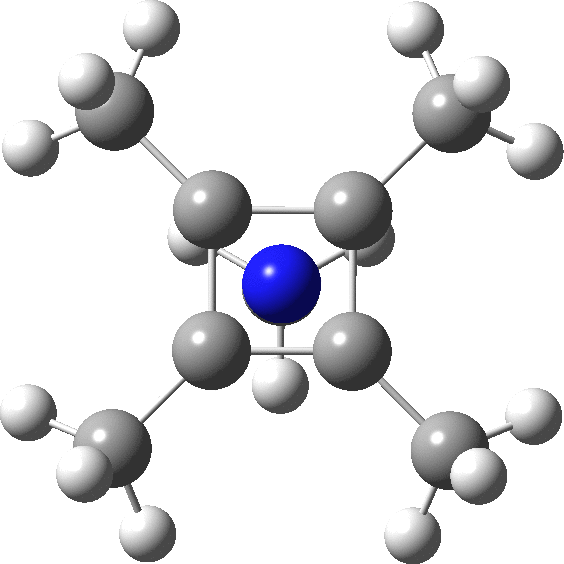
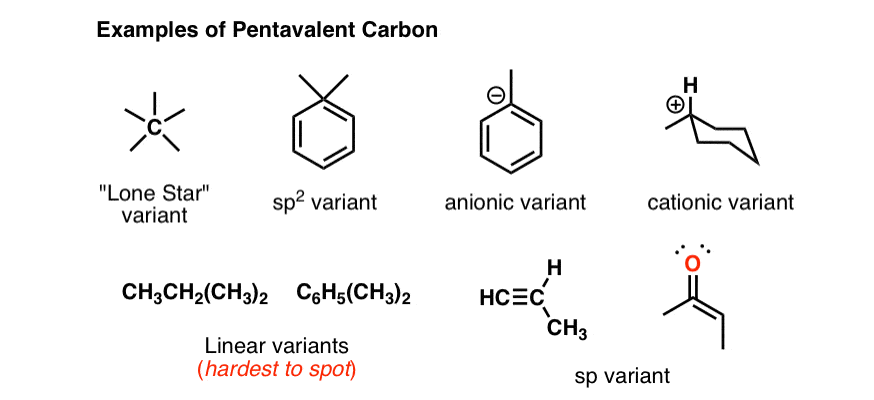
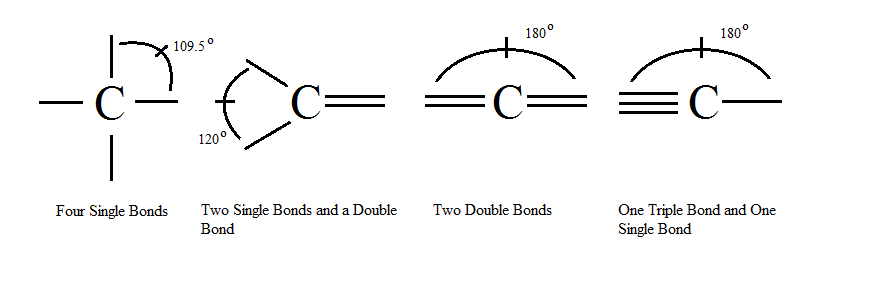carbonyl compounds - Can a carbon that has 5 bonds be explained by saying that one of its bonds is a coordination bond? - Chemistry Stack Exchange
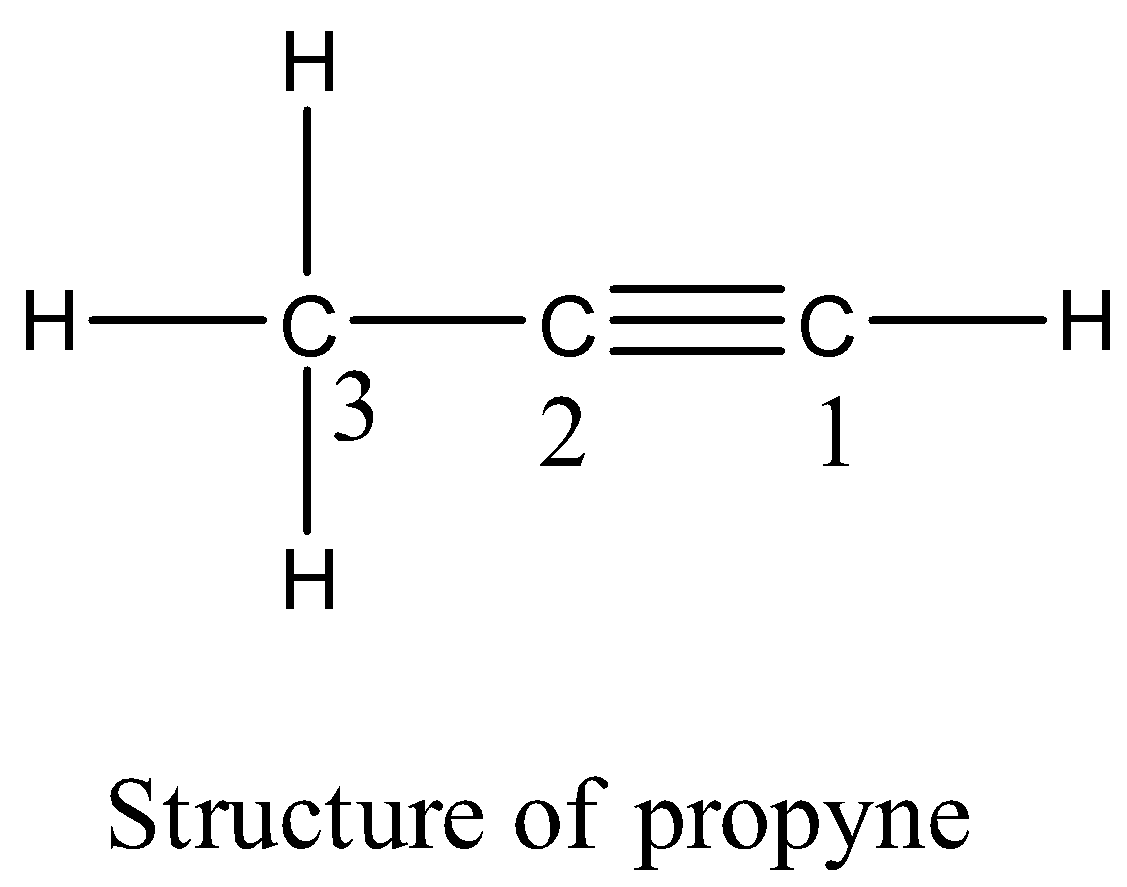
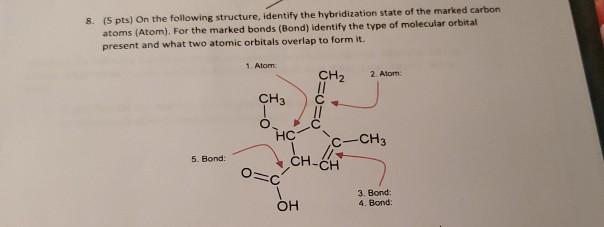
Propyne molecule contains:A. 6 sigma and 2 pi bondsB. 5 sigma bondsC. 5 pi bonds and 1 sigma bondD. 2 sigma and 3 pi bonds

Name a 5-carbon chain with a double bond on the 1st carbon and a hydroxyl substituent on carbon 3. | Homework.Study.com






:max_bytes(150000):strip_icc()/texas_carbon-56a12ddd5f9b58b7d0bcd225.png)
.gif)