
Materials | Free Full-Text | Nanoporous Anodic Aluminum-Iron Oxide with a Tunable Band Gap Formed on the FeAl3 Intermetallic Phase

XRD pattern of iron aluminum oxide nanoparticles for oxidation time 5... | Download Scientific Diagram

This rock has a composition of 60% Aluminum Oxide, 20%Moissanite, 16% Mullite, and 4% Aluminum Iron Oxide III. What is it and where did it come from? I've talked to foundry specialist

SOLVED: When aluminium metal reacts with iron (III) oxide a single displacement reaction occurs to form iron and aluminum oxide. Using the Standard Enthalpy Chart write the balanced chemical equation and calculate
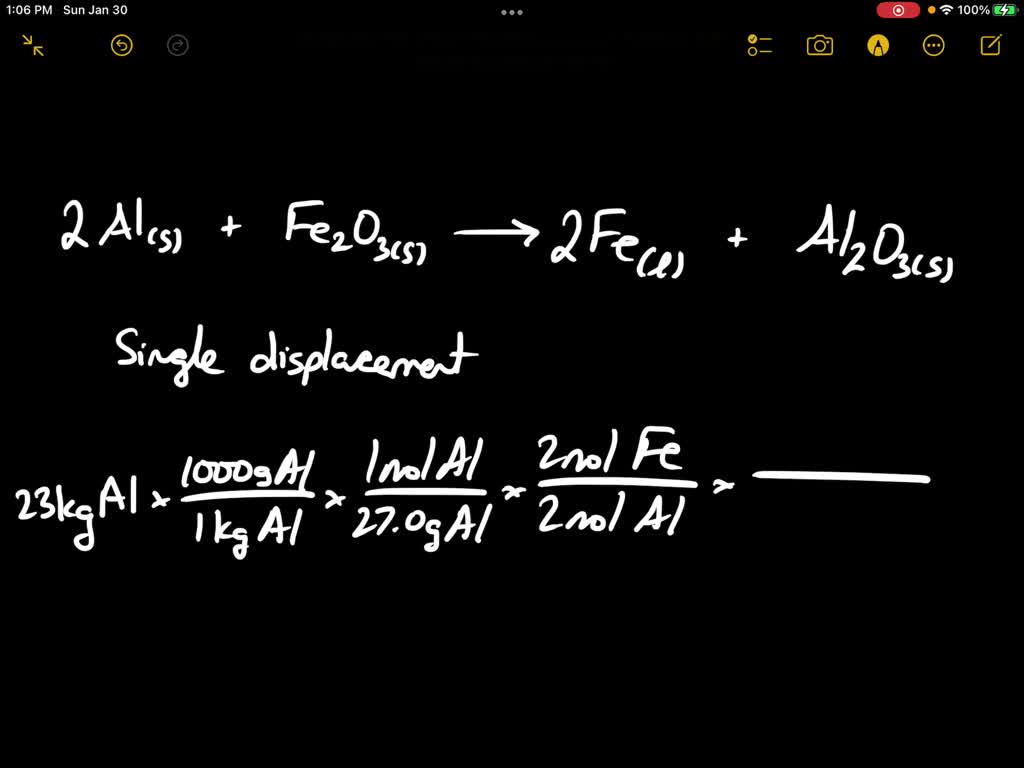
SOLVED: Solid aluminum reacts with solid iron (III) oxide to produce liquid iron and aluminum oxide powder. Write a balanced chemical equation for this reaction showing the reactants and products. Include subscripts
![Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download](https://images.slideplayer.com/33/8235959/slides/slide_4.jpg)
Thermite By Jack Rjeili. What is Thermite? Mix of aluminum powder and iron oxide [1] Once ignited, produces extreme temperatures Produced from union. - ppt download

when a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and - YouTube
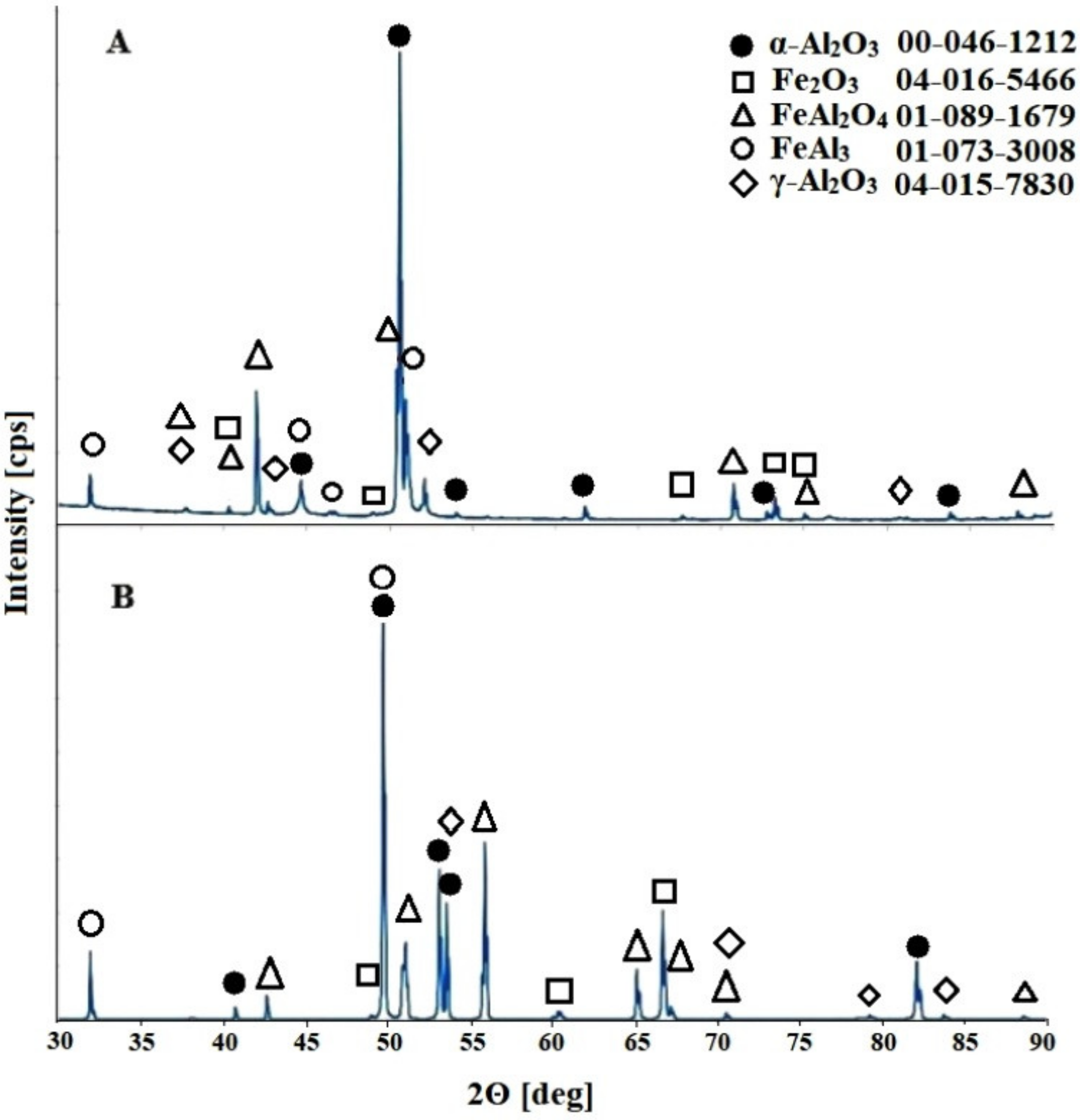
Materials | Free Full-Text | Nanoporous Anodic Aluminum-Iron Oxide with a Tunable Band Gap Formed on the FeAl3 Intermetallic Phase
![PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/035f93a77120a19b7cb355ec122296f5319649ce/3-Figure1-1.png)
PDF] Mechanism for thermite reactions of aluminum/iron-oxide nanocomposites based on residue analysis | Semantic Scholar

Clay minerals, iron/aluminum oxides, and their contribution to phosphate sorption in soils — A myth revisited - ScienceDirect













