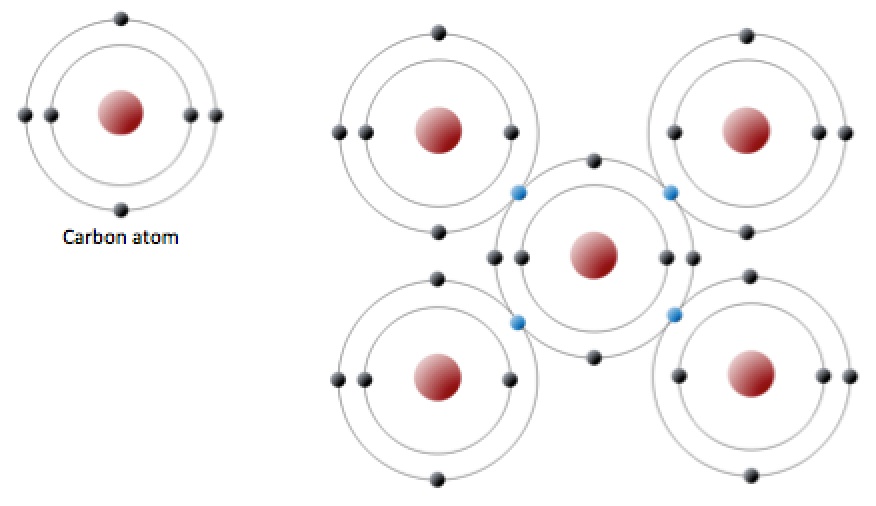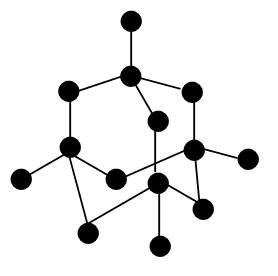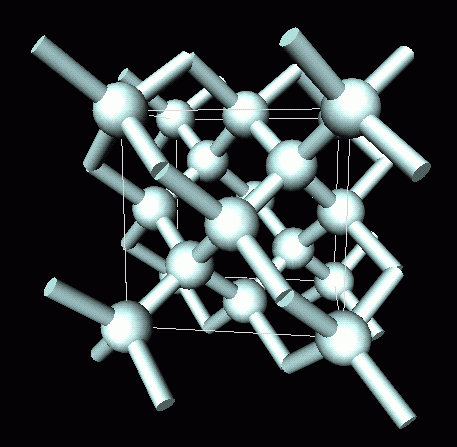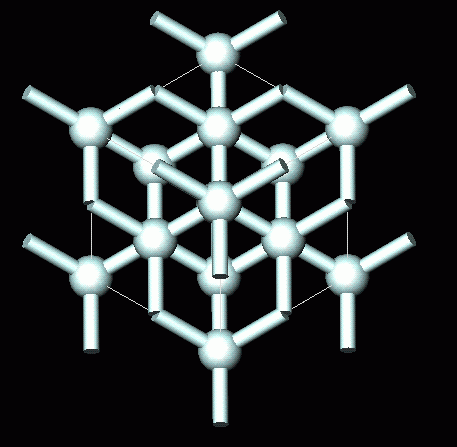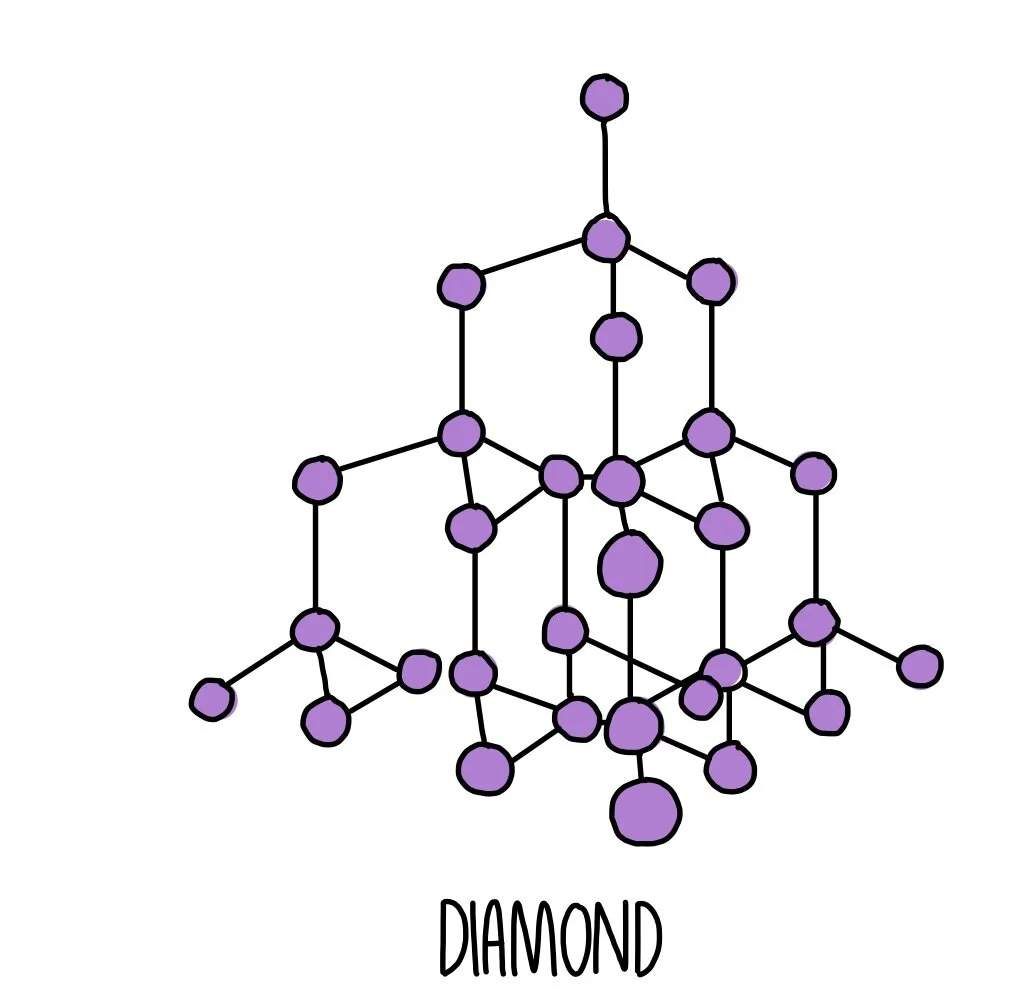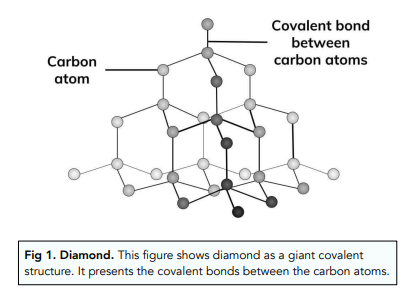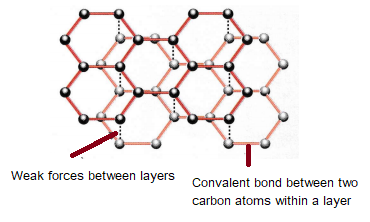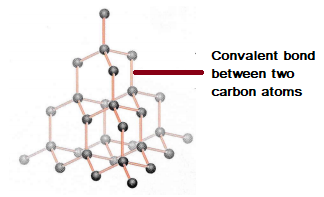
GCSE CHEMISTRY - What is the Structure of Diamond? - What is the Structure of Silicon? - GCSE SCIENCE.
Why diamond is still the hardest material and what keeps us away from developing harder material than diamond? - Quora

When non-metals combine together they share electrons to form molecules A covalent bond is a shared pair of electrons Non-metal + non-metal → Covalent. - ppt download

Crystallographic structure of diamond with tetrahedral bond angles of... | Download Scientific Diagram
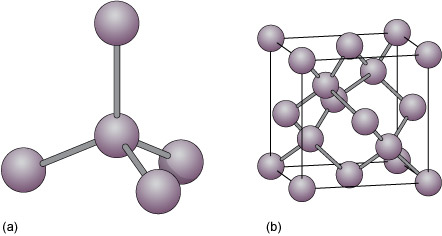
Minerals and the crystalline state: 4.3 Covalent structures and bonding - OpenLearn - Open University