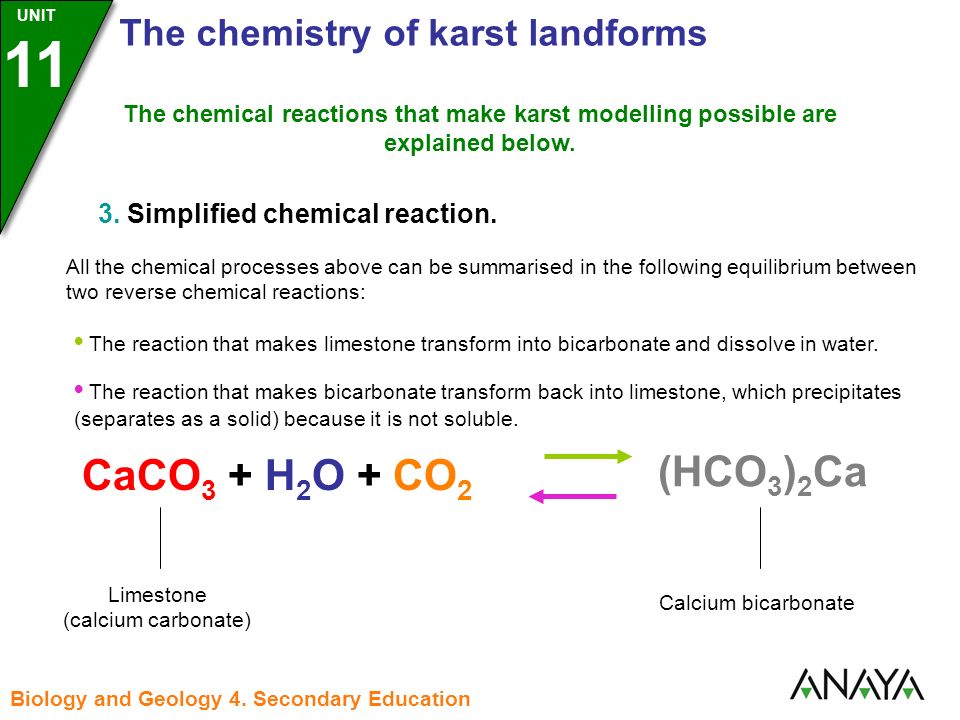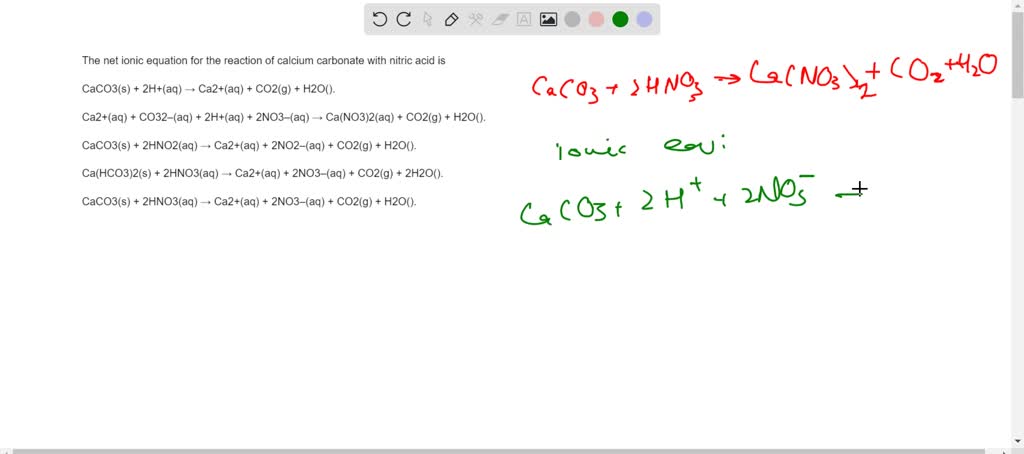
SOLVED: The net ionic equation for the reaction of calcium carbonate with nitric acid is CaCO3(s) + 2H+(aq) → Ca2+(aq) + CO2(g) + H2O(). Ca2+(aq) + CO32–(aq) + 2H+(aq) + 2NO3–(aq) →

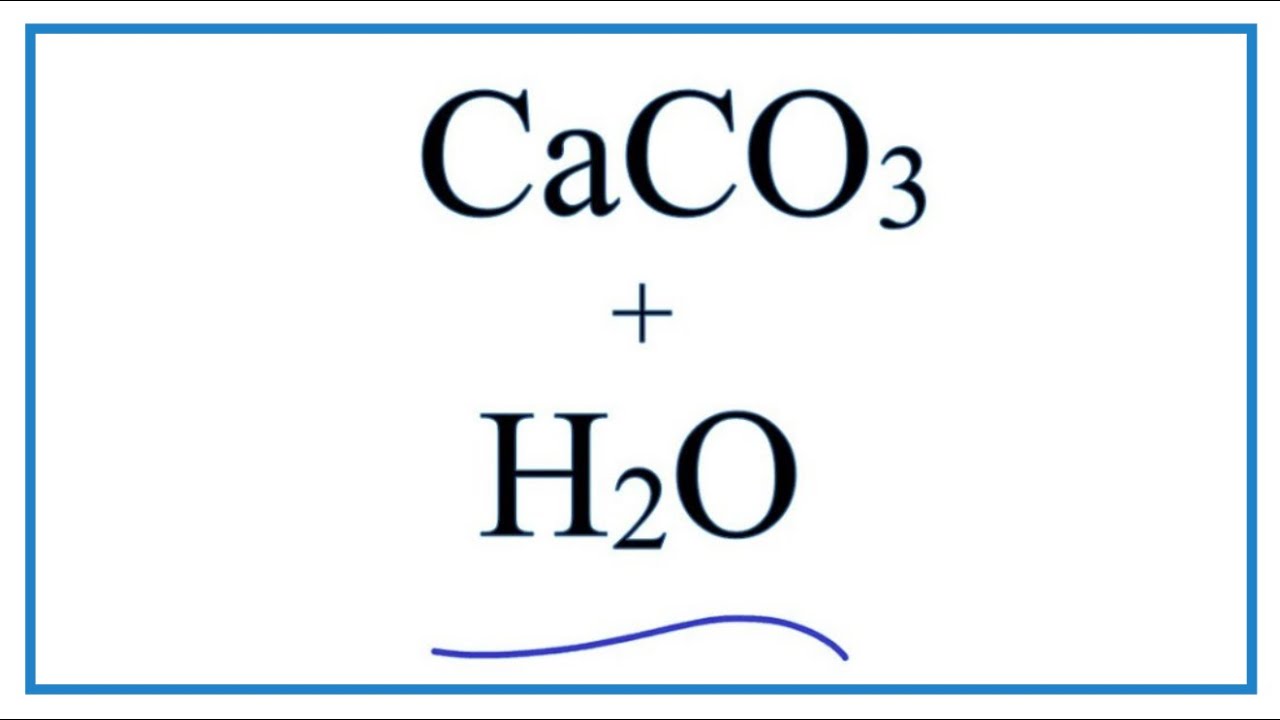
How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O | How to Balance CaCO3 + HCl = CaCl2 + CO2 + H2O Hey there! Are you struggling with balancing
Q. Calcium carbonate reacts with aqueous HCL to give Calcium chloride and carbon dioxide ,according to the reaction CaCo3(s)+2HCl(aq) →CaCl2(aq)+CO2(g)+H2O(l) What mass of calcium carbonate is required to react completely with 25

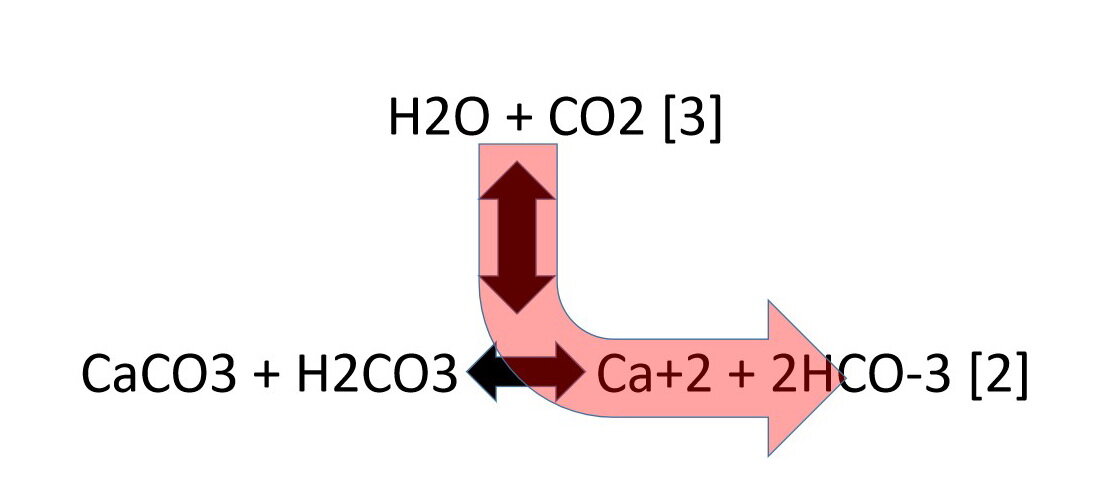
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram
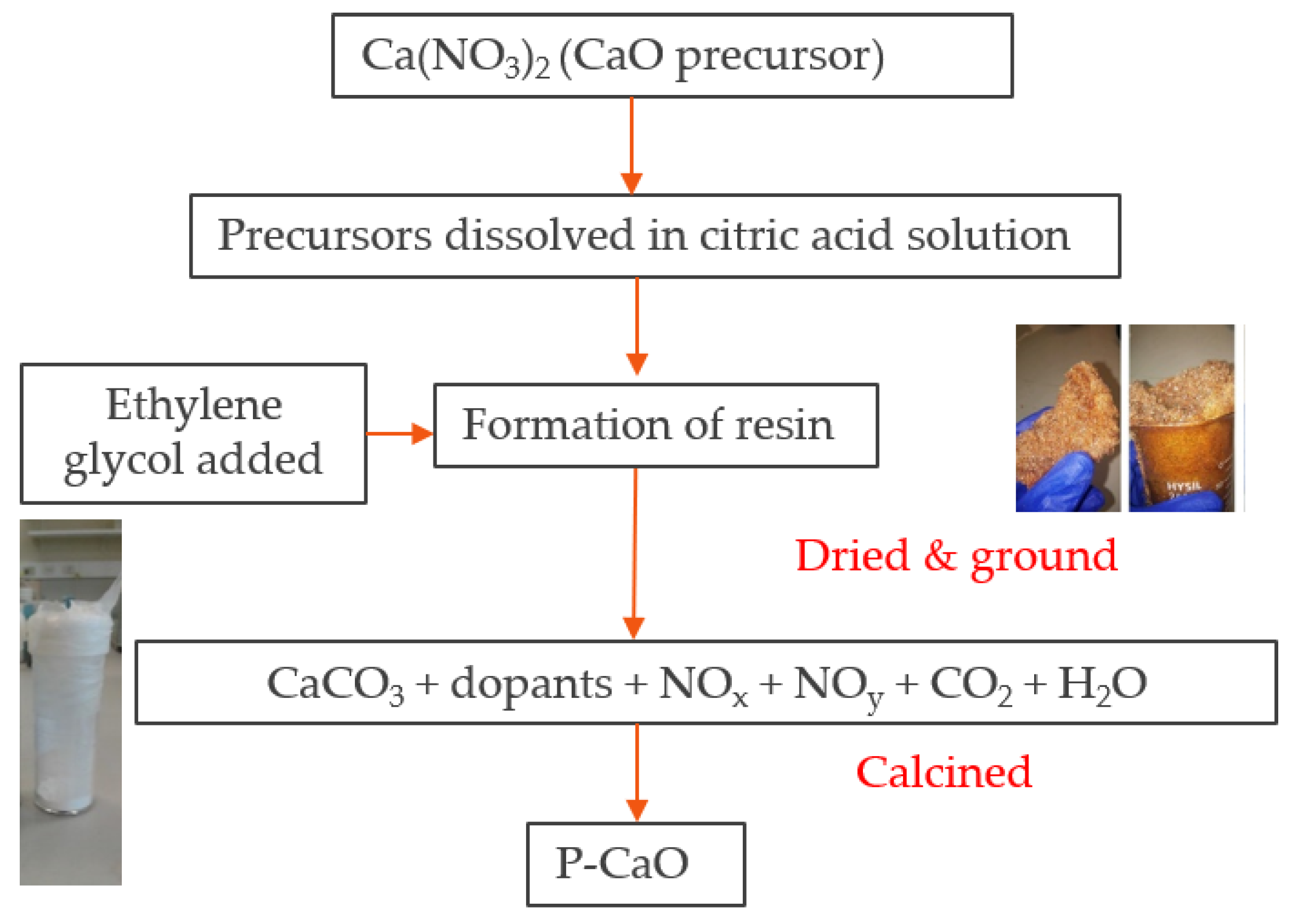
Applied Sciences | Free Full-Text | Comparative Kinetic Analysis of CaCO3/CaO Reaction System for Energy Storage and Carbon Capture

100 g CaCo3 reactwith20g HCl according tobfollowing equation CaCo3 +2HCl=CaCl2+H2o+Co2 What is mass of Co2 willbeproduced - Chemistry - Some Basic Concepts of Chemistry - 12831553 | Meritnation.com