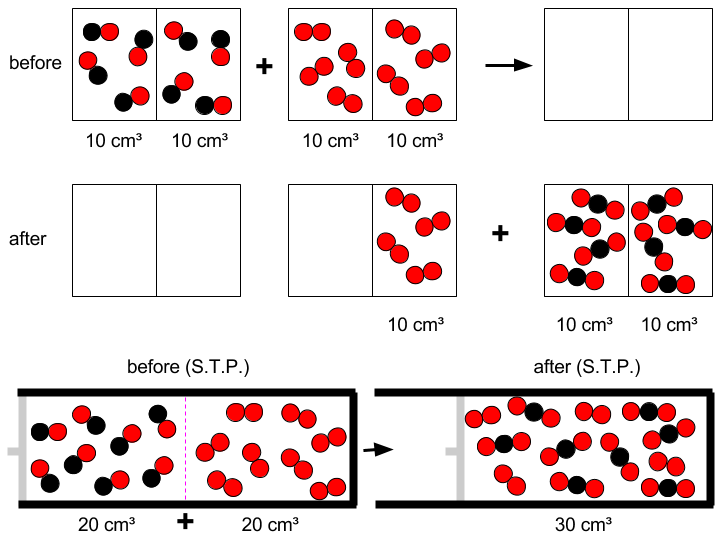
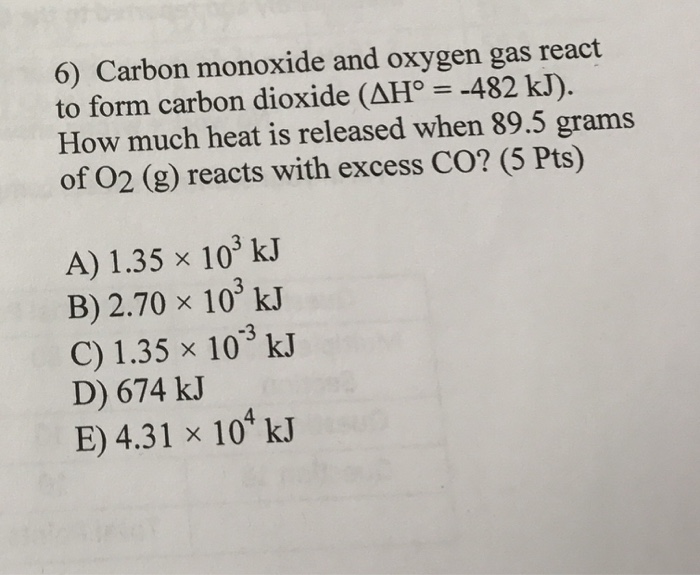Explosion limits of the dry carbon monoxide + oxygen reaction - Transactions of the Faraday Society (RSC Publishing)

Carbon reacts with oxygen to produce carbon dioxide which is an exothermic reaction, where 394 kJ/mol of heat is produced. What will be the value of Δ H when 24 grams of

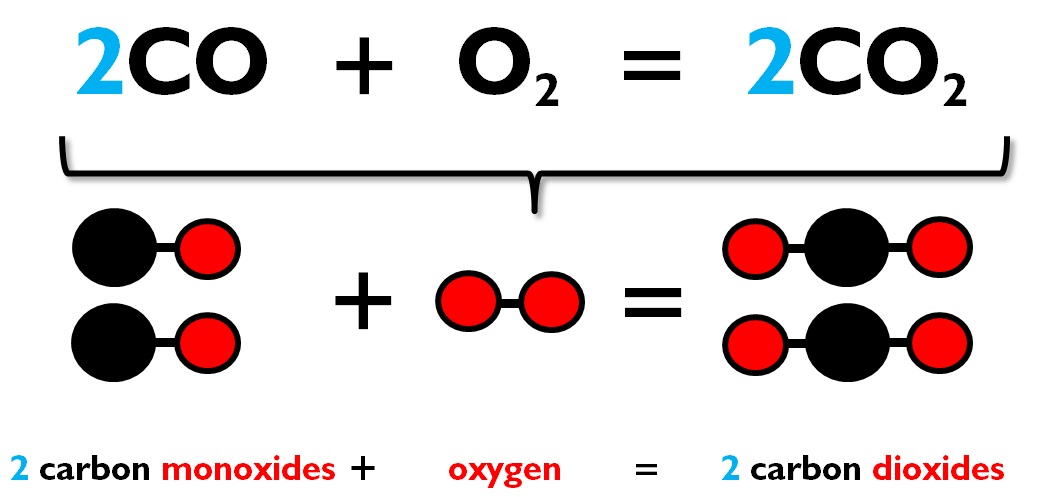
SOLVED: 19. Carbon monoxide reacts with oxygen according to the equation below: 2 cO(g) + Oz(g) v 2 COz(g) What is the overall order of the reaction? a. first-order b second-order third-order
![SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism](https://cdn.numerade.com/ask_images/736b473b932d4d048614f30fae3d5333.jpg)
SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism
.png)
how will carbon react with oxygen what happens when the product is dissolved in water show along with - Science - Materials Metals and Non-Metals - 1217495 | Meritnation.com

SOLVED: A) The following represents the equilibrium reaction between carbon monoxide, oxygen and carbon dioxide: 2C0 (g) + O2 (g) 7 2CO2 (g) A container is filled initially with CQz, CQ and