electrochemistry - Electrolysis with salt water on steel plates creates horrible black liquid - Chemistry Stack Exchange

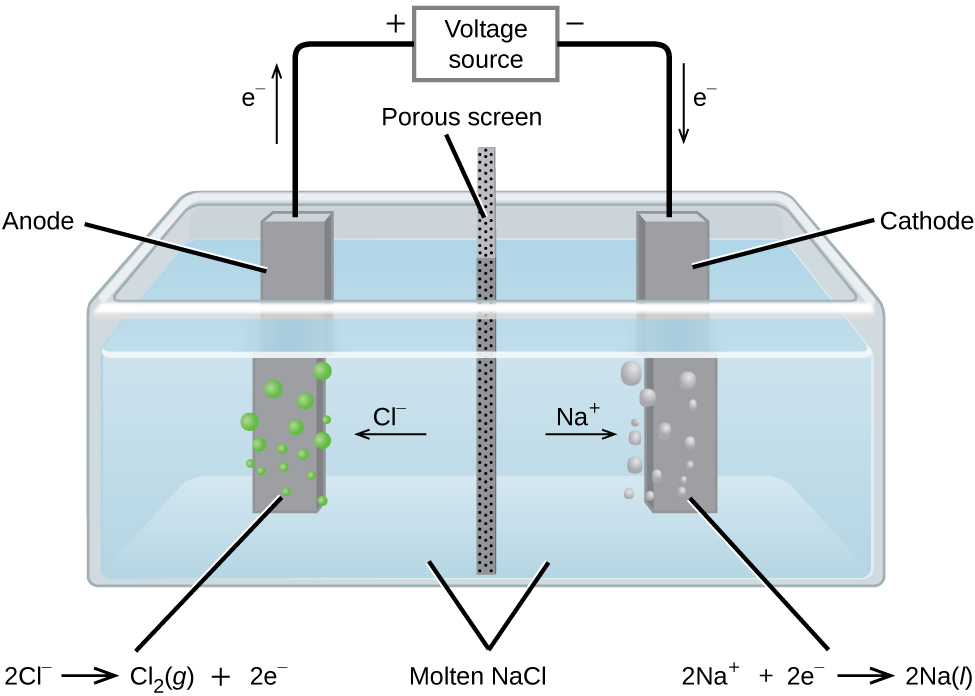
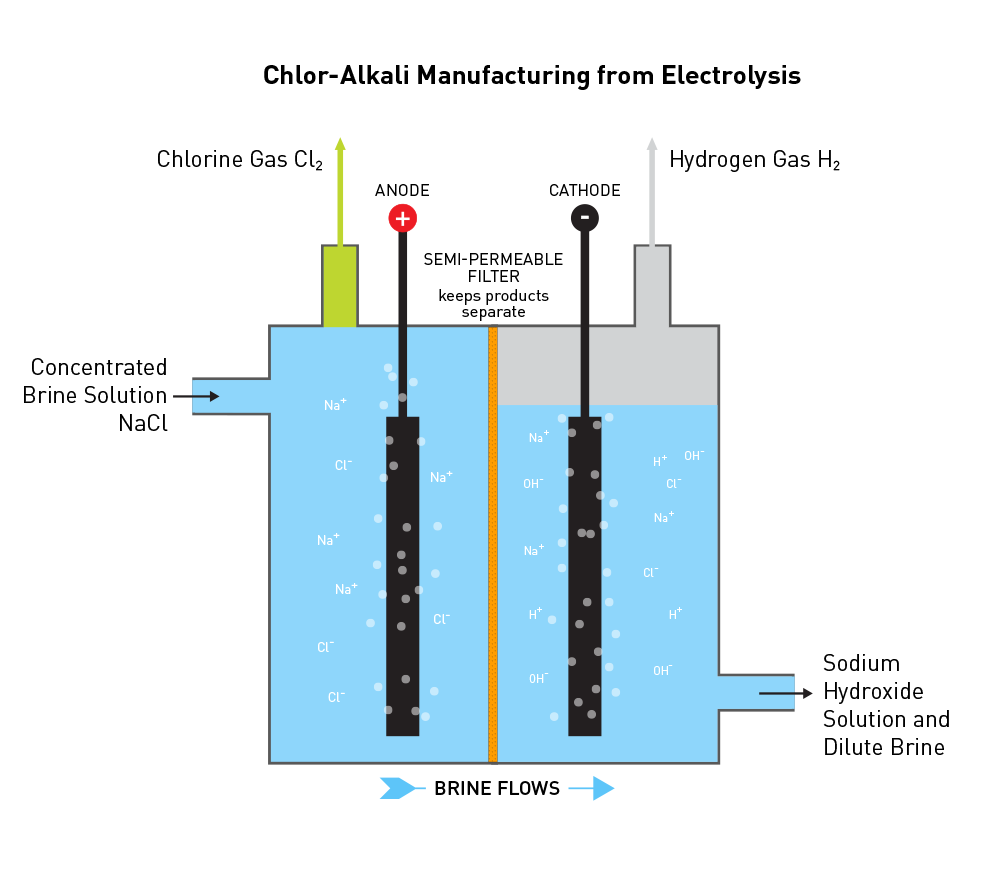
In the electrolysis of aqueous sodium chloride (NaCl), what are the half-cell reactions that occur at each electrode? (refer to image) | Homework.Study.com
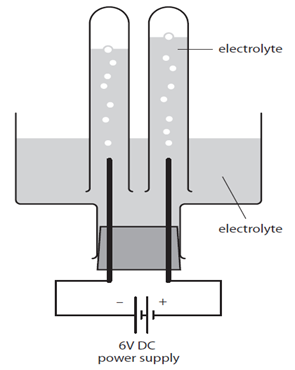
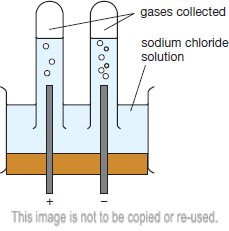
1:60 (Triple only) practical: investigate the electrolysis of aqueous solutions - TutorMyself Chemistry

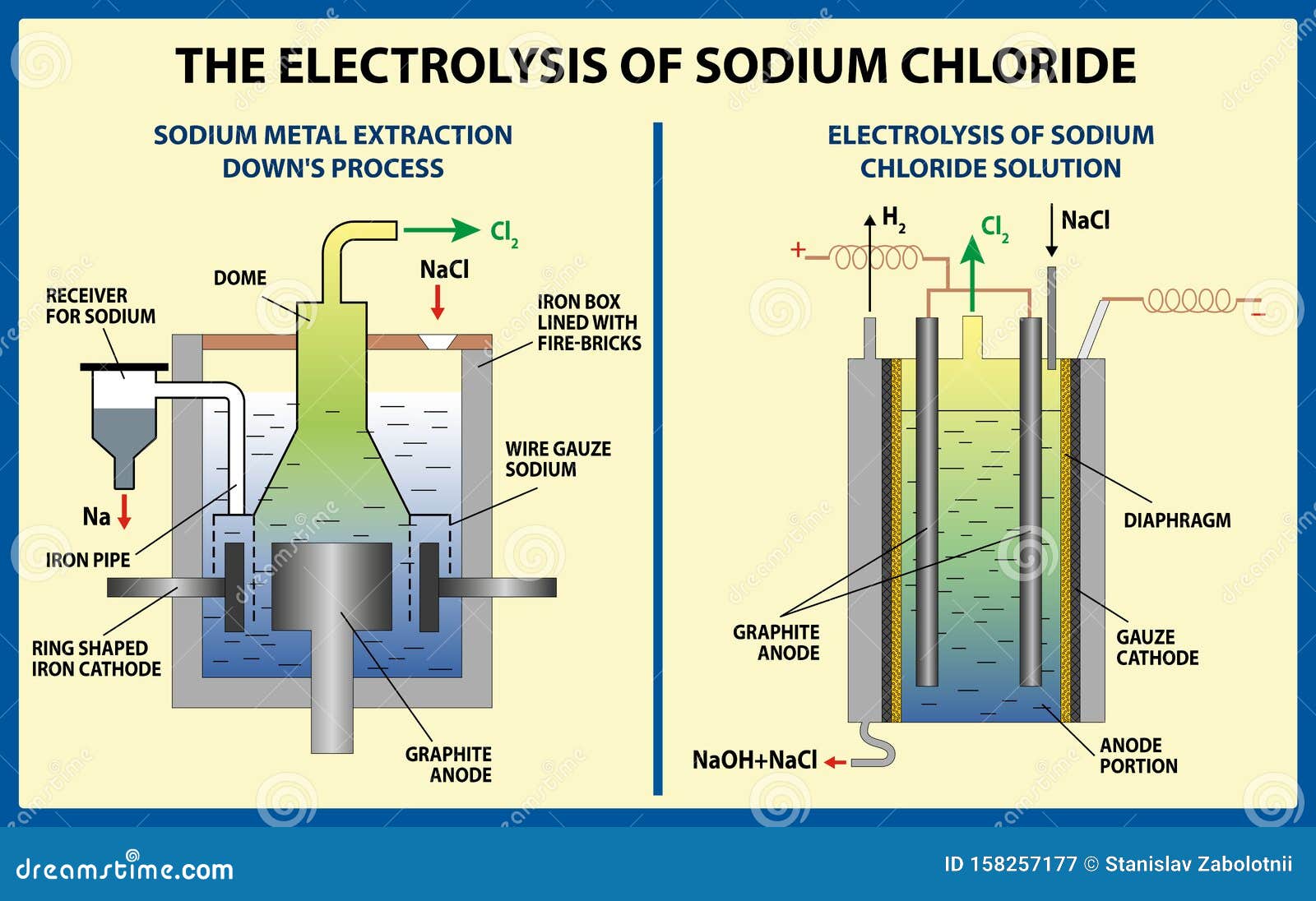
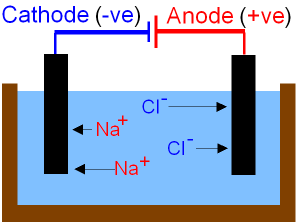
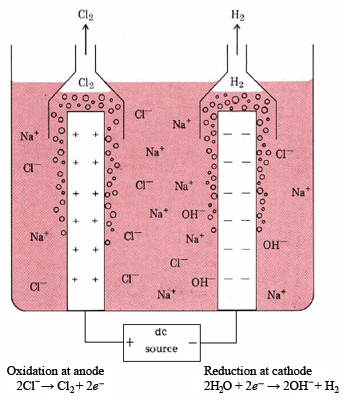
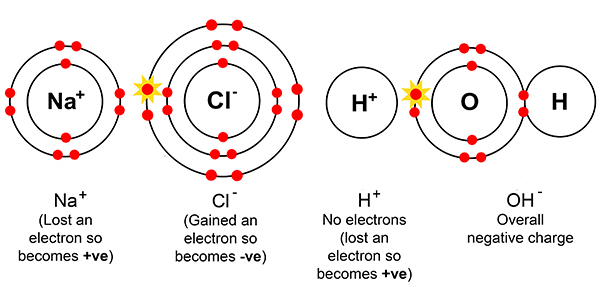
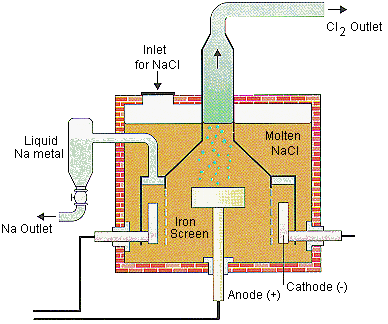
electrolysis of sodium chloride solution brine product equations electrodes anode cathode apparatus electrolyte cell sodium bromide potassium iodide gcse chemistry KS4 science igcse O level revision notes

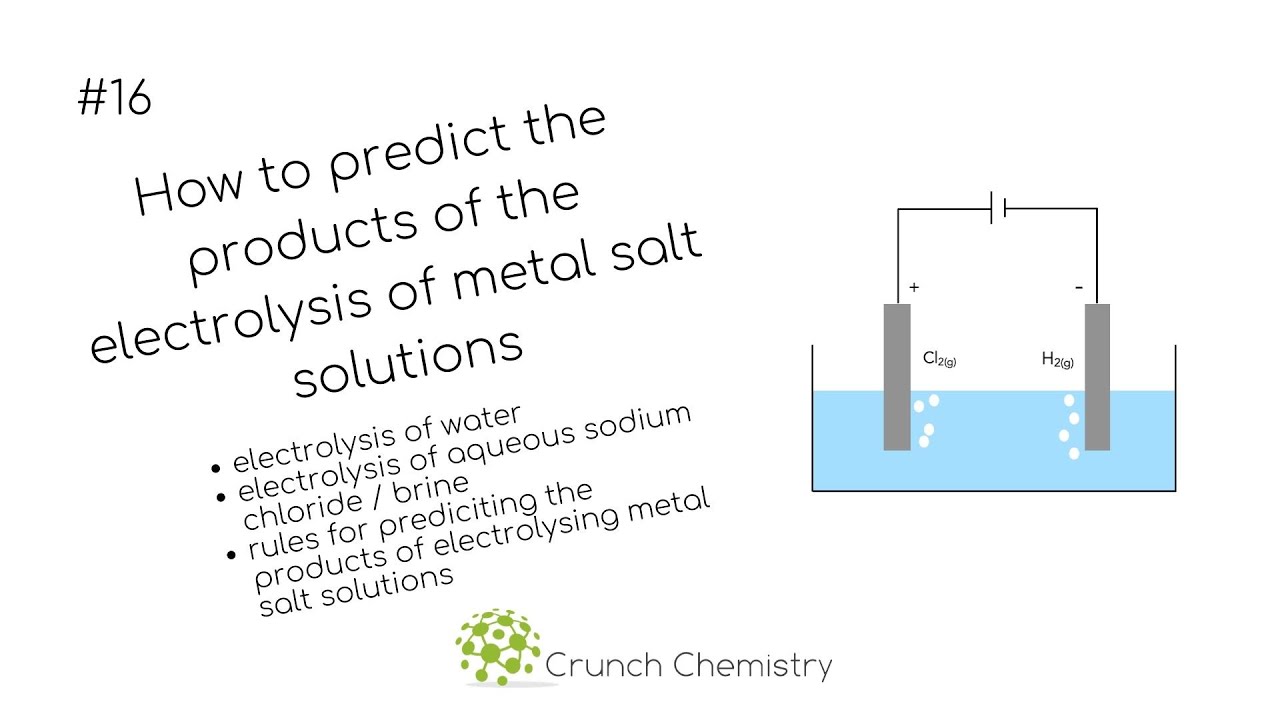
If I add table salt (NaCl) into water and do electrolysis do I get chlorine and hydrogen gases? No oxygen gas at all? - Quora

electrolysis of sodium chloride solution brine product equations electrodes anode cathode apparatus electrolyte cell sodium bromide potassium iodide gcse chemistry KS4 science igcse O level revision notes