
Question Video: Identifying Which Metal Can React with Acid and Its Oxide Can Be Reduced by Carbon | Nagwa

SOLVED: ACTIVITY SERIES OF METALS IN AQUEOUS SOLUTIONS most active (easily oxidized readily lose electrons) lithium potassium barium calcium sodium These metals displace hydrogen from water: Ki Ba Ca Na Cas 2H,Ow




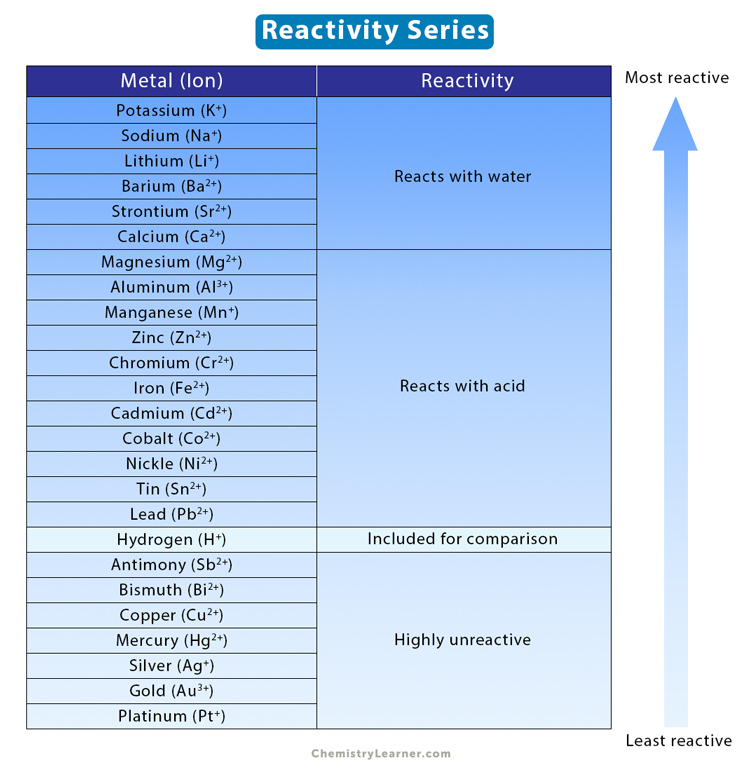










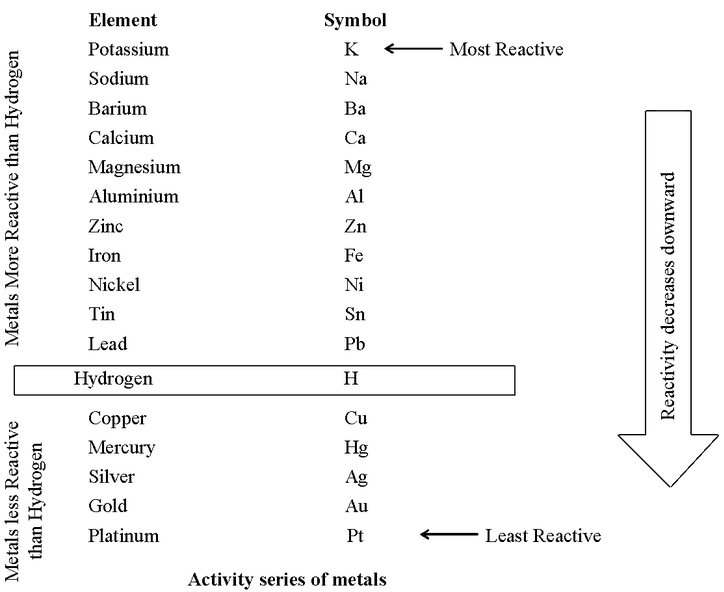


![Reactivity Series of Metals - Chart [and How to remember] - Teachoo Reactivity Series of Metals - Chart [and How to remember] - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/b0a28f61-1fb3-456e-8ae8-e110e8999f99/reactivity-series-01.jpg)

