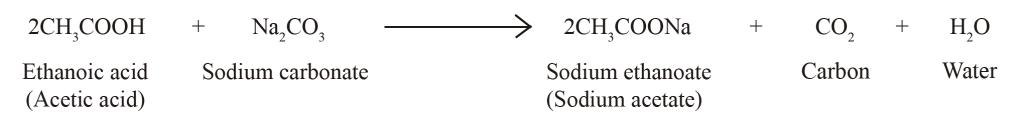Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora

10.5 Equilibria in Salt Solutions LO: I understand how the pH of a salt solution comes about. - ppt download

Sodium Chemicals - Sodium Hydroxide, Sodium Chloride, Sodium Acetate Anhydrous And Edta Di Sodium Salt Supplier & Distributor
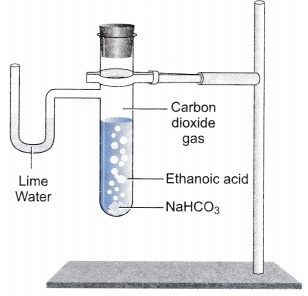
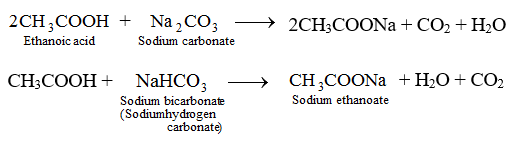
A salt X is formed and a gas is evolved when ethanoic acid reacts with sodium hydrogen- carbonate - CBSE Class 10 Science - Learn CBSE Forum
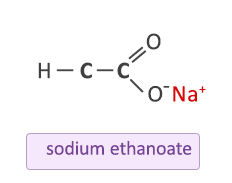
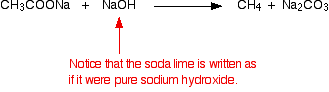
Write three different chemical reactions showing the conversion of ethanoic acid to sodium ethanoate. - Sarthaks eConnect | Largest Online Education Community

Buffers. A buffer is a solution whose pH is resistant to change on the addition of relatively small quantities of an acid or base. Buffers have the ability. - ppt download