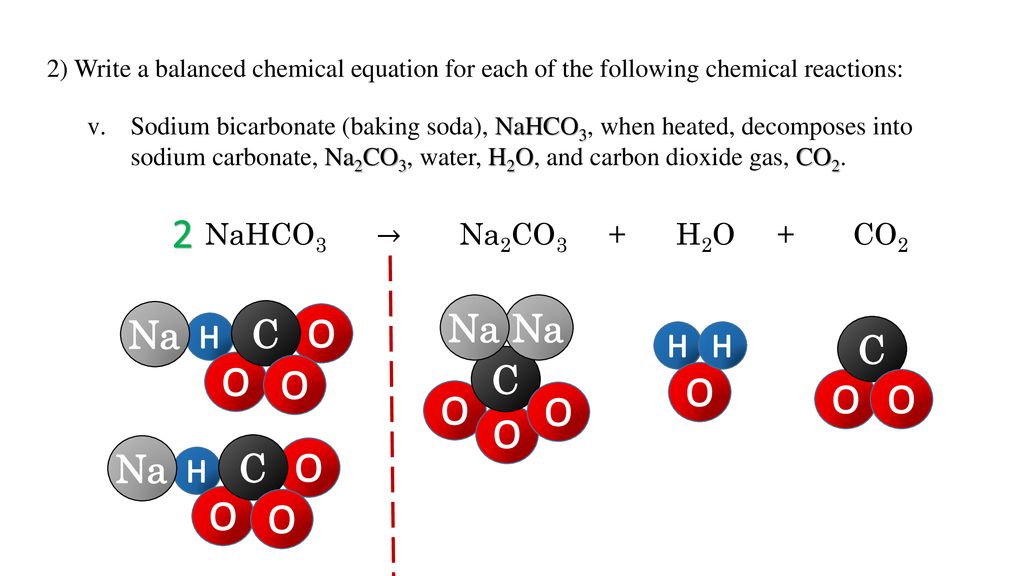
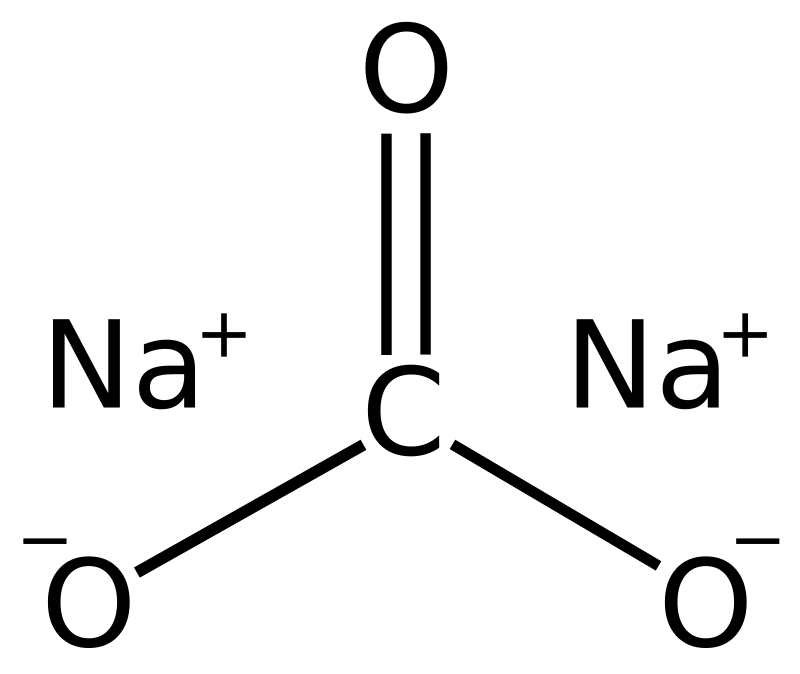
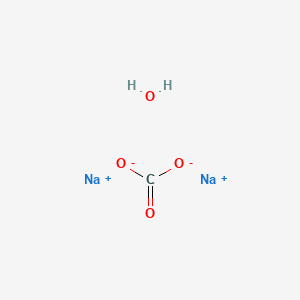
_how-to-balance-nahco3-naoh-na2co3-h2o-sodium-bicarbonate-sodium-hydroxide.jpg)
How to Write the Net Ionic Equation for ZnSO4 + Na2CO3 = ZnCO3 + Na2SO4 from znco3 naoh Watch Video - HiFiMov.co

Phase Equilibria of the NaOH–NaBO2–Na2CO3–H2O System at 30 °C, 60 °C, and 100 °C | Journal of Chemical & Engineering Data

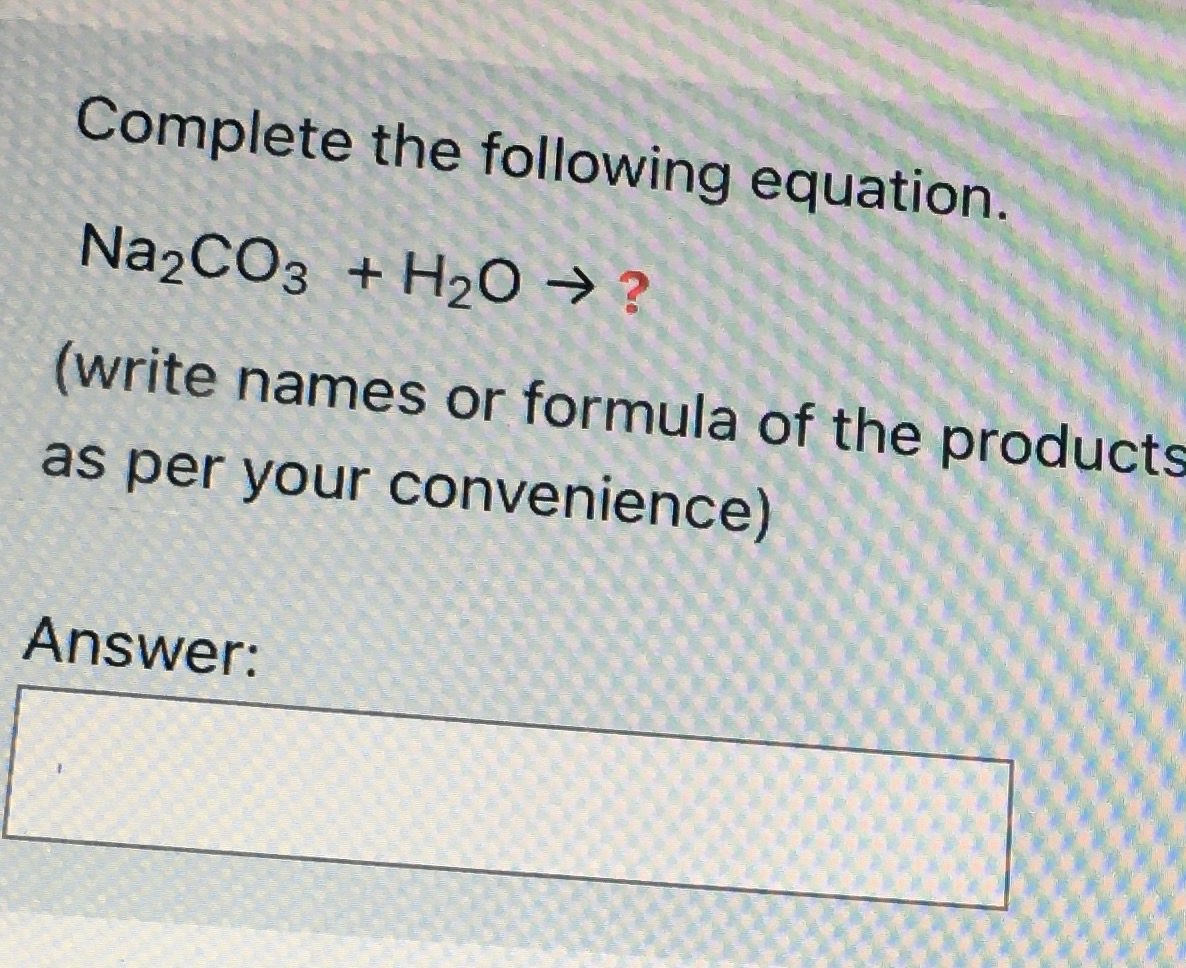
How to Balance Na2CO3+H2O→NaOH+CO2 | How to Balance Na2CO3+H2O→NaOH+CO2 | By Organic Chemistry Tutorial/Inorganic Chemistry/Science - Facebook

This chemical equation represents a chemical reaction. Na2CO3 + 2HCl - 2NaCl + CO2 + H20 Which chemical is - Brainly.com
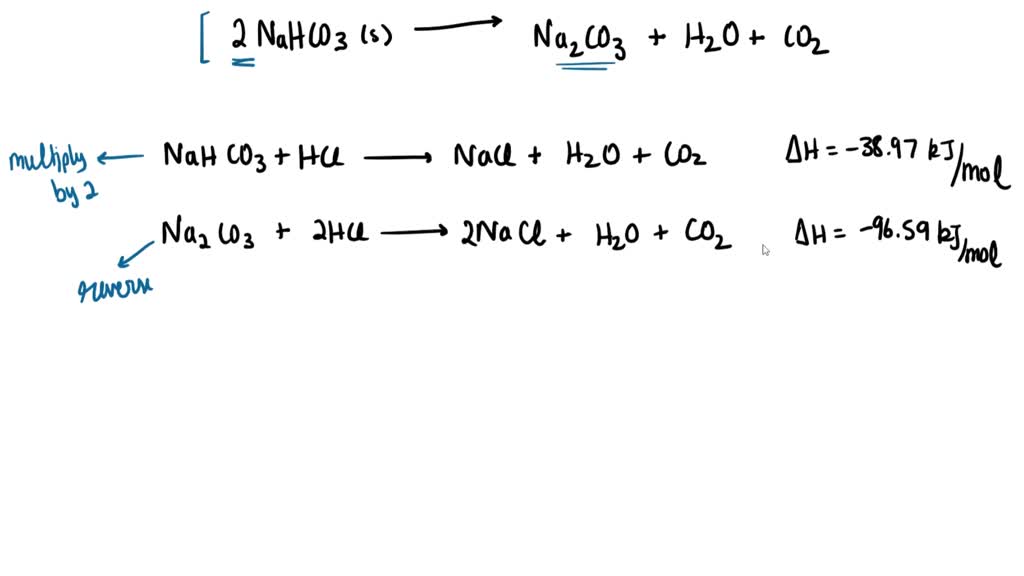
SOLVED: For the reaction Na2CO3 (s) + 2HCl(g) → NaCl(s) + CO2 (g) + H2O(l) ∆H is -144.1 kJ. What is ∆U.
What volume of 0.25M Hcl is required to react completely with 22.6g of Na2co3 according to the equation Na2Co3+2Hcl=2Nacl=H20 (2) The molecular mass of organic compound is 78
_how-to-balance-na2co3-hcl-nacl-h2o-co2-sodium-carbonate-hydrochloric-acid.jpg)