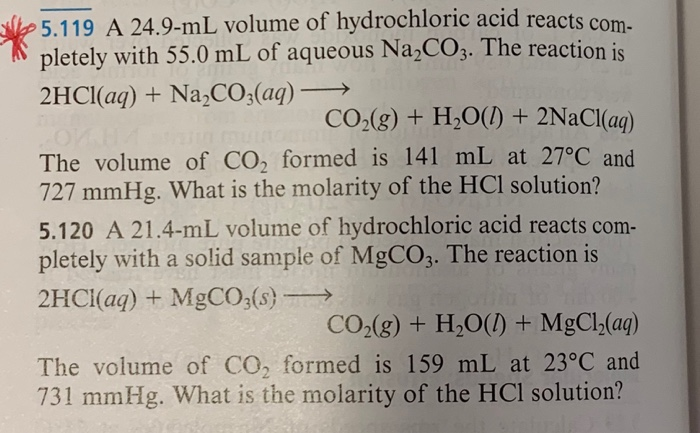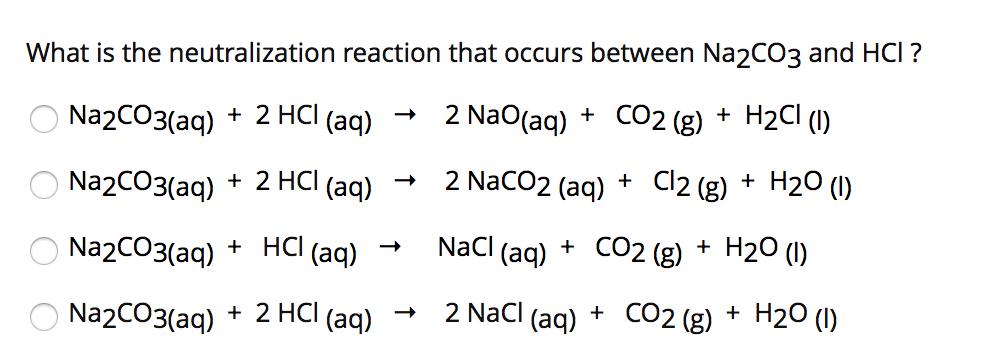25.3 g of sodium carbonate, Na 2 CO 3 is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, the molar concentration of sodium ion, Na ^+

Question Video: Identifying the Observations of the Reaction between Hydrochloric Acid and Sodium Carbonate | Nagwa

Question Video: Determining the Products Formed from the Reaction between Sodium Carbonate and Hydrochloric Acid | Nagwa
in the titration of Na2CO3 by HCl using methyl orange indicator, thevolume required at the equivalence point will be if that of the acidrequired using phenolphthalein indicator is 10.0 ml:
What will be the mass of sodium chloride formed when 5.3 g of sodium carbonate is dissolved in 250 ml of a half molar HCl solution? - Quora

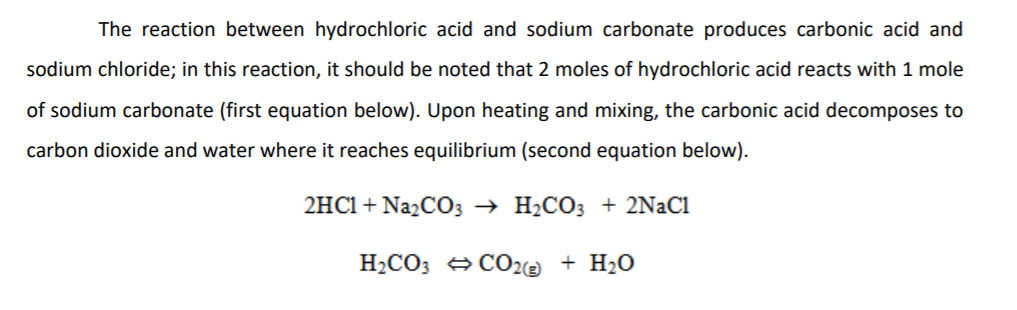
Net Ionic Equation for Na2CO3 + HCl | Sodium Carbonate + Hydrochloric Acid | Net Ionic Equation for Na2CO3 + HCl | Sodium Carbonate + Hydrochloric Acid Hello, Chemistry Enthusiasts! For today's
0.1 g of a solution containing Na2CO3 and NaHCO3 requires 10 ml, of 0.01 N HCl.neutralization using phenophthalein .wt
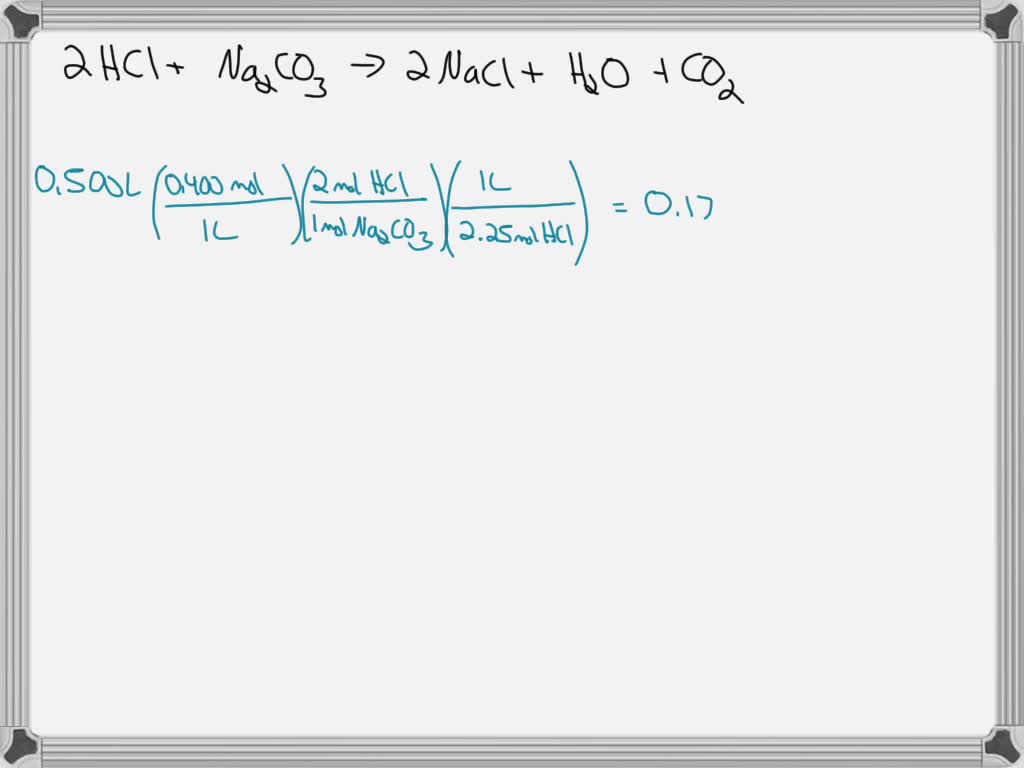
SOLVED: HCL reacts with NaCO3 forming NaCl waters and carbon dioxide this equation is balanced as written: 2HCl (aq) + Na2 CO3(aq) right arrow 2NaCl (aq) +H2O (l) + CO2)g) 1- what
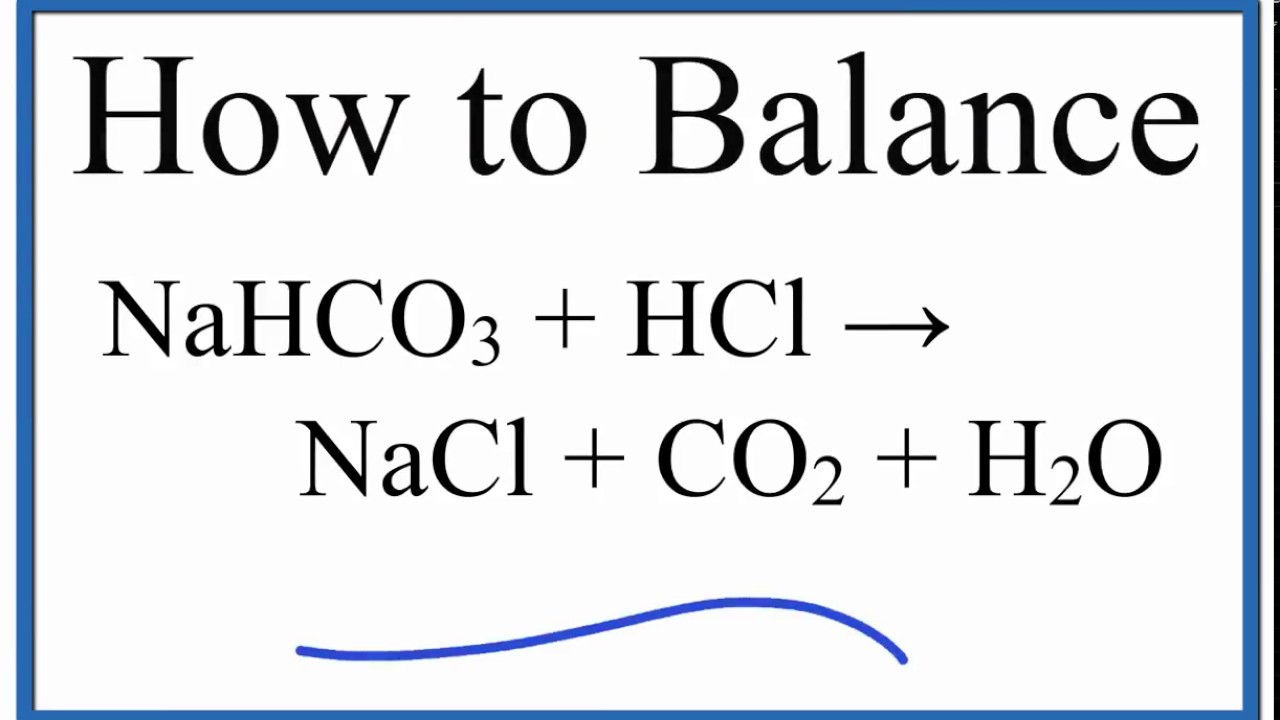
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube