Handbook of carbon, graphite, diamond, and fullerenes : properties, processing, and applications | Semantic Scholar
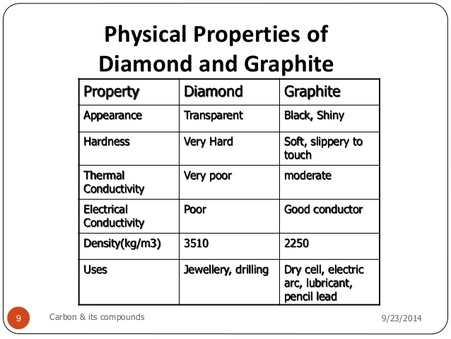
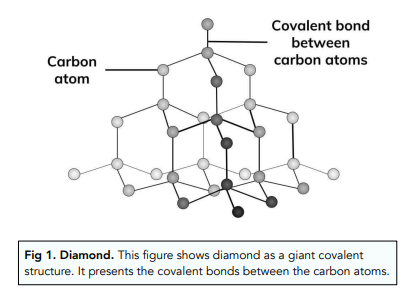
Explain the difference in properties of diamond and graphite on the basis of their structures. - Sarthaks eConnect | Largest Online Education Community
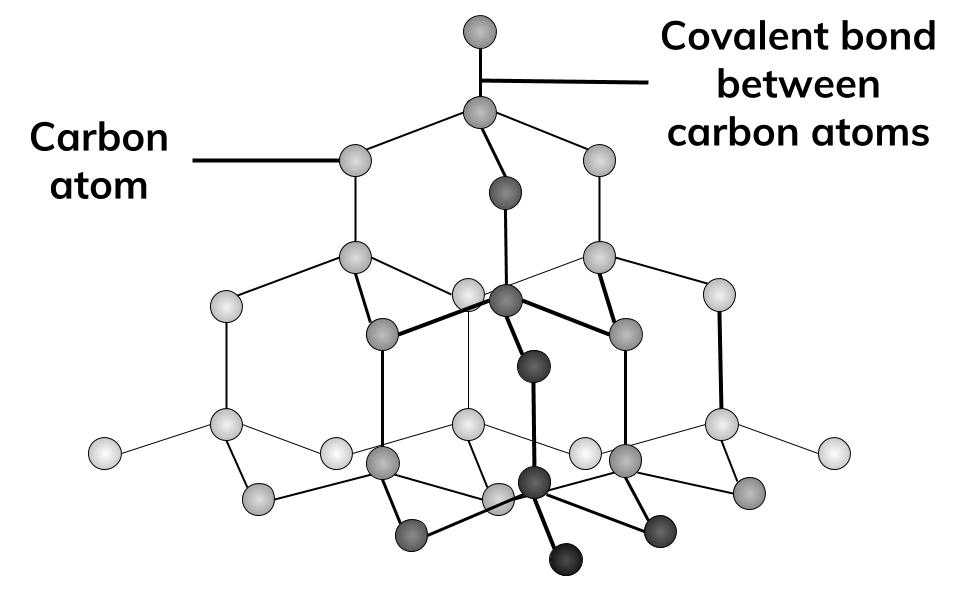
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry